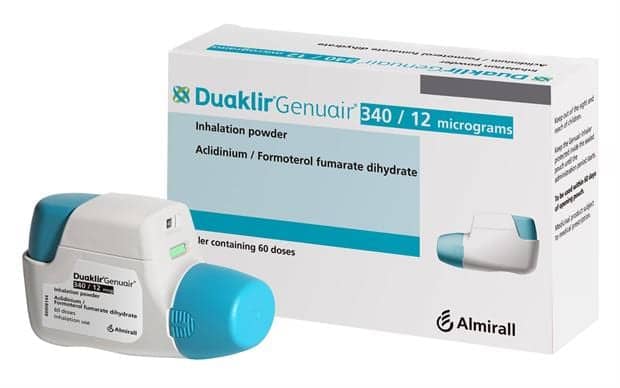
AstraZeneca Canada has announced that the COPD treatment system DUAKLIR GENUAIR is now available in Canada and through the Canadian public health system.
DUAKLIR is an approved fixed-dose LAMA/LABA combination of two long-acting bronchodilators — aclidinium bromide, a long-acting muscarinic antagonist (LAMA), and formoterol fumarate, a long-acting beta-agonist (LABA) — administered through the GENUAIR device, which research results have found patients preferred when compared to other devices. The inhaler is composed of a pre-loaded, multi-dose dry powder inhaler (DPI) that results in efficient delivery of the drug throughout the lungs. Furthermore, because the GENUAIR device contains two verification modes (‘click’ sound and color indicator change), patients with COPD are reassured that they received the correct administration of the daily doses.
In summary, DUAKLIR GENUAIR treatment system helps in reducing frequency of daily COPD respiratory symptoms. This, in turn, eases patients’ anxiety as well as possible stress of dosage/symptom related issues.
“Despite the treatment options available to patients living with COPD, there is still a significant unmet need when it comes to daily symptom control,” says Dr. Ronald Grossman, respirologist, Credit Valley Hospital and professor of medicine at University of Toronto. “DUAKLIR GENUAIR provides patients with a safe, effective way to improve breathlessness over 24 hours. This improves lung function and gives them the confidence that their dosage is working consistently throughout the entire day.”