Small, lightweight, and easily portable, handheld spirometers are becoming more ubiquitous in the industry. But can they expand the reach of early spirometry into primary care settings?
It’s no secret that spirometry testing is underutilized,[1] despite its effectiveness at diagnosing chronic lung diseases like COPD and asthma. Spirometry is a crucial tool in diagnosing but also managing these diseases.[1] While the majority of respiratory therapists are well-versed in spirometry and pulmonary function testing, the consistent call remains for greater use and access to the technology in primary care settings.
A 2016 study[2] assessed the accuracy of diagnoses and utilization of spirometry in multiple primary care clinics. After review, researchers found only 29% of patients had spirometry testing or had results available and 31.6% of the patients diagnosed with COPD were misclassified because the spirometric measurements did not confirm the disease.[2] A 2015 study of primary care clinics revealed that only 19% of patients with COPD had undergone spirometry testing.[3] After an education-based intervention called “WipeCOPD,” which included monthly continuing education modules and live spirometry training sessions for primary clinicians, those same clinics’ utilization rates rose to 56% and then 82% within nine months.[3] The lack of use is not limited to adult patients; according to a 2010 study in Pediatrics, only 21% of pediatricians and primary care doctors routinely used spirometry to establish an asthma diagnosis, classify severity, and assess childrens’ level of asthma control—the three clinical situations outlined in the national asthma guidelines.[4]
Of course, this is about more than technology adoption. According to ndd Medical, early diagnosis of COPD, along with ongoing treatment and monitoring, can slow the progression of lung disease and have a significant impact on the quality of a patient’s life.[5] But as noted above, first-line clinicians in primary care settings need more access to and training with simple, effective spirometers. As noted by Philips Healthcare, a a major cause is lack of awareness and training, which may cause physicians to avoid spirometry and diagnose COPD strictly based on symptoms.[6] However, access to portable handheld spirometry can move PFT beyond the lab and into primary care, where physicians are able to detect airway obstruction earlier and initiate faster treatment, ndd explained. In fact, researchers report that screenings by primary care doctors using office spirometry can increase early diagnoses in COPD patients by twofold and they recommend these clinicians get their hands on quality spirometers.[7]
ON THE MARKET
MicroDirect offers the MicroLoop, an advanced handheld spirometer that includes 41 test parameters with % predicted, diagnostic interpretation, lung age, choice of predicted normals, child incentives, post bronchodilator comparison and printed report with graphs via optional PC software.[8] MicroLoop employs Micro Medical’s precision Gold Standard Digital Volume Transducer which is especially suited to measuring the very low flow rates seen in patients with COPD, MicroDirect explained. The device’s Digital Volume Transducer is an extremely stable form of volume transducer, which measures expired air directly at BTPS (Body Temperature and Pressure with Saturated water vapor) thus avoiding the inaccuracies of temperature corrections. The transducer is insensitive to the effects of condensation and temperature and avoids the need for individual calibration prior to performing a test, the company said. According to a comparison of 10 handheld spirometers,[7] the MicroLoop was one of only three devices that had acceptable limits of agreement for FVC and one of four devices that had acceptable limits of agreement for FEV1.
An additional option from MicroDirect is the Micro 1 Spirometer, which is specifically designed for situations where low cost precision spirometry measurements are required. The Micro 1 Spirometer is extremely lightweight and portable, making it suitable for physician offices, hospital outreach clinics, bedside screening, health fair screenings and other situations where remote testing is required, the company said.[8]
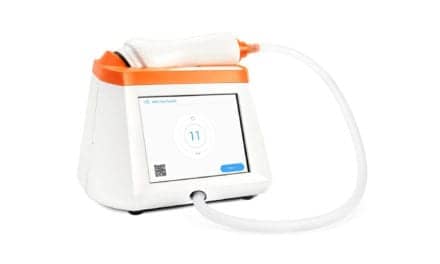
ndd Medical Technologies Inc released its newest spirometry solution this year, the EasyOne Air.[9] The handheld device is designed for primary care physicians, who are typically the first line for detecting chronic lung diseases like COPD. In addition, EasyOne Air fully satisfies the Global Initiative for Obstructive Lung Disease (GOLD) guidelines. Powered by ndd’s ultrasound flow technology called Ultrasonic TrueFlow, EasyOne Air anticipates interfering variables, allowing it to function independently of gas composition, pressure, temperature and humidity, ensuring consistent, standardized test results every time. The portable and connected device requires no calibration, warm-up time or maintenance and provides clinicians with a simple and proven method of lung function testing. EasyOne Air’s full-color touch screen display also ensures accurate and immediate results through real-time curves and parameters. The device connects (via USB) to a computer for direct printing and downloading while also offering secure Bluetooth connection. The device offers seamless integration with patients’ electronic medical records through EasyOne Connect, a platform for all EasyOne point-of-care solutions.[9]
Vitalograph’s micro handheld spirometer is a reliable, affordable spirometry option. The compact, lightweight, handheld spirometer includes a full-color touchscreen, icon driven menu, and highly accurate and robust flow sensor technology.[10] The device features a Fleisch Pneumotachograph, a diagnostic flow measurement technology offers a wide range of advantages, including highly accurate and reliable measurements of very low flows and very high flows.
In addition, the In2itive spirometer combines the benefits of full spirometry, data integration and mobility in a small lightweight, handheld spirometer. Fleisch flow measuring technology and a simple icon driven menu, plus a large, high resolution color touchscreen and storage capacity for 10,000 subjects, makes In2itive an ideal solution for high quality spirometry on the move. Manual entry of subject data is optional as In2itive makes it possible to select all subject data from Spirotrac Software and download it automatically, saving time and eliminating data entry errors, as per the company.[10]
Medical International Research (MIR) offers three versions of its Spirobank II handheld spirometer: Smart, Basic, and Advanced.[11] All three models offer pre/post-bronchodilator comparisons, full reports, and memory for up to 10,000 spirometry tests. Spirobank II Smart and Spirobank II Advanced are equipped with oximetry options, while the Spirobank II Basic is ideal for family doctors, primary care, occupational medicine, screening. The Spirobank II Smart comes with a Virtual Assistant via iPad compatibility, which supports the operator before, during and after the spirometry test, according to MIR.
Also available from MIR is the Spirodoc, which is equipped with various operation modes: “advanced” parameters for the specialist, “reduced” set of parameters for screening as well as a “simplified” version for home care operation. The device also features SpO2 oximetry that can be used for six-minute walk tests and sleep analysis, according to the company.
Futuremed’s Spiropalm is a handheld version of the company’s Discovery-2 spirometer. The device is a more portable, economical option for primary care or specialists and meets or exceeds ATS standards for accuracy.[12] The device offers bronchial challenge testing and also includes a pediatric incentive called “Bubbles,” which encourages patients to perform tests properly so you have optimal results quickly and easily. Its unique sensor design is not affected by environmental changes. The built-in temperature sensor and the ability to make BTPS corrections ensure that measurements are exact for a wide range of flows, with low resistance. RT
For more information, contact [email protected]