In the past, clinicians felt that CPAP was boring and nontechnical, but with newer technology, CPAP has been found to be an innovative modality dominating neonatal nurseries around the world.

CPAP involves applying positive pressure to the airway of a spontaneously breathing patient. It works, in the treatment of patients with unstable lung mechanics, by maintaining inspiratory and expiratory pressures at levels that are above ambient pressure. CPAP increases functional residual capacity and decreases airway resistance. This process decreases work of breathing, improves static lung compliance, stabilizes minute ventilation, and allows the delivery of a greater tidal volume for a given pressure.
In preterm infants with poor respiratory mechanics, CPAP prevents the collapse of the distal airways, and this improves ventilation. Recently, at Thomas Jefferson University Hospital, Philadelphia, the neonatal team including MDs, RNs, and RCPs discussed becoming more aggressive in the use of CPAP in order to promote early extubation and prevent intubation in the delivery room.
Stabilizing the Airways
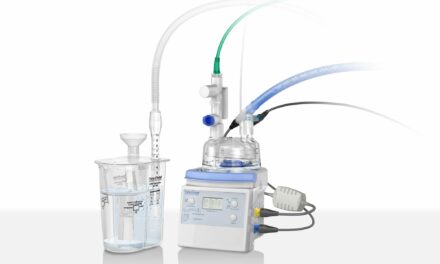
The team’s first step was to look at CPAP delivery devices that could ensure proper stabilization of the airways. The CPAP devices already in use at the institution were not adequate for the care of very small premature neonates; the units used narrow nasal prongs which caused patients to have problems exhaling through them due to increased resistance from narrow prongs and continuous-flow from the ventilators, which affected patient synchrony adversely. Conventional CPAP created a problem because it forced the infant to exhale against the incoming gas, which increases the work of breathing. Using nasopharyngeal prongs and conventional CPAP ventilators worked reasonably well for infants larger than approximately 1,500 g, but newer technologies were tried with the delivery of CPAP with smaller infants in mind. A device having separate channels for inspiratory and expiratory gas is now in use; the system helps with the movement of gas in and out of the neonate’s lungs.
A disposable component of the system causes the direction of the gas to change with the infant’s breathing patterns while maintaining a constant pressure throughout the respiratory cycle. When the patient inspires, the gas takes the path of least resistance and is directed to the patient’s airways. Upon expiration, the gas again takes the path of least resistance and goes out an exhalation tube, reducing resistance during the exhalation phase. The key to the functionality of the CPAP generator is this unique action, which occurs within the generator in harmony with the infant’s breathing pattern. The internal design of the generator allows it to manipulate the gas flow to provide a stable CPAP level at the patient’s airway.
Because the device depends on this fluidic-flip mechanism, not valves, its response to the patient’s respiratory effort is extremely rapid. Gas is sent to the infant when he or she needs it; during expiration, the effect of the fluidic flip is active exhalation. The fluidic flip is based on the Coanda effect (a stream emerging from a nozzle tends to follow a nearby curved surface, if the curvature of the surface or angle at which the surface meets the stream is not too sharp). Fluid (O2/air) is entrained or drawn in by the momentum of the flow. If there is an obstruction, there is less fluid to be drawn in and there is a decrease in pressure on one side of the jet as a result. The decrease in pressure causes a deflection in the flow and redirects the jet until it sticks to the wall. The CPAP generator uses the Coanda effect by having the infant’s breathing effort trigger the fluidic flip inside the device.
Prime Candidates
Once the team had chosen a CPAP device with which everyone was comfortable, it started to consider which patients would most benefit from CPAP. Prime candidates were those of low birth weight, and those who had been extubated, had failed to breathe normally without assistance, and had then been reintubated.
The team decided to use CPAP as a first-line treatment for respiratory distress syndrome (RDS) in the ICU and in the delivery room. RDS was seen most frequently in the delivery room at the birth of premature patients. After birth, premature infants such as those with low birth weight or with RDS were intubated, given a dose of surfactant, and extubated; then, CPAP treatment was started. The patients were stabilized and transported to the intensive care nursery while using CPAP because the team felt that it was important to recruit and stabilize the airways (and not to disrupt the CPAP level).
Concerns
There were some concerns about using the newer devices to deliver CPAP, since the nasopharyngeal prongs of the older models were easy to use. It was very simple to extubate the patient, place the prongs in his or her nares, and then use the ventilator already at the bedside to provide continuous flow. The newer machine must be brought to the bedside. In addition, there are new techniques to learn for affixing the prongs and hat to the patient properly. Since the team instituted a more aggressive approach and learned to use the new device, it has been very impressed with the results, which included decreased intubation and less reintubation.
When RCPs go to the delivery room, they are conscious of peak inspiratory pressures; they tend to begin treatment with mask CPAP and let the patients predict their outcomes. If the patient is breathing, we do not intubate because of their size or gestational age. MDs tend to wait longer to intubate patients with apnea or high Paco2s. After surfactant has been administered and patients have been stabilized in the delivery room, they are moved without discontinuation of CPAP.
Pressures of 5 to 7 cm H2O are used, usually with an initial fraction of inspired oxygen (Fio2) of 40%. The patient is weaned from supplemental oxygen aggressively, with the Fio2 set to maintain oxygen-saturation levels of 88% to 92%. The Fio2 typically is reduced to 25% to 30% within the first few hours of treatment and maintained at a lower level.
Lower Intubation Rates
Subjectively, the team seems to be seeing a lower intubation rate and fewer extubation failures. It is very exciting to be able to walk through the NICU and see more CPAP machines and fewer intubated patients. RCPs are busier, however, because there is a lot of work involved in preventing intubation. RCPs were not accustomed to transporting patients who were using CPAP, and they were not in the habit of affixing straps and bonnets. CPAP patients are also checked more often to avoid skin and nasoseptal breakdown.
In the past, some clinicians felt that CPAP was uninteresting and nontechnical, but with newer technology, it has become an innovative modality dominating neonatal nurseries around the world.
Raymond Malloy, RRT, is director of pulmonary care, Thomas Jefferson University Hospital, Philadelphia.
References
1. Moa G, Nilsson K, Zetterstrom H, Jonsson LO. A new device for administration of nasal continuous positive airway pressure in the newborn: an experimental study. Crit Care Med. 1988;16:1238-1242.
2. Klausner JF, Lee AY, Hutchison AA. Decreased imposed work with a new nasal continuous positive airway pressure device. Pediatr Pulmonol. 1996;22:188-194.