|
The care of premature infants in neonatal intensive care units (NICUs) began in the late 1960s, and it has been one of the most significant contributions toward reduction of perinatal mortality. The early efforts to improve patient survival were directed to decrease cyanosis and improve ventilation. Oxygen administration improved cyanosis, but it was later recognized to contribute to retrolental fibroplasia, now known as retinopathy of prematurity (ROP).1 In the 1970s, improvement in technologies, such as oximetry monitoring and newborn ventilation, enabled clinicians to provide respiratory care for newborn infants.
As the birth rate of very low birth weight (VLBW) infants increases, health care providers are faced with providing adequate oxygen to support recovery without causing long-term complications. The goal is to provide enough oxygen to prevent central nervous system (CNS) hypoxia, organ tissue hypoxia, and ductal patency. On balance, too much oxygen may lead to lung tissue injury, eye injury, and oxidative stress. Compounding this situation is the presence of lung disease, and immature lungs, eyes, and antioxidant system. In addition, there is a need for frequent FiO2 manipulation to treat inherent transient hyperoxemia.
Oxygen Toxicity
High levels of oxygen can cause oxidative stress (damage to the myocardium and kidney) and negatively impact survival. Gladstone et al showed a correlation between oxygen use and an increase in protein-bound carbonyl in lung fluid, which is a marker for oxidative injury.2 In addition, Munkeby et al demonstrated that oxygen at high concentrations, even for short periods of time, can produce a significant increase in inflammatory markers.3 Some infants could be predisposed to chronic lung disease (CLD) as demonstrated in a study by Tsao et al, which showed a direct correlation in placenta growth factor (P1GF) levels in cord blood at birth and risk for premature infants to develop CLD, also known as bronchopulmonary dysplasia (BPD).4
Delivery Room
The concept of lung protective strategies, including minimal tidal volume, a lower change in pressure, and minimal oxygen therapy to prevent ventilator-induced lung injury (VILI), is very familiar to NICU clinicians.5,6 Although these interventions are commonly used, clinicians appear less aware that the same approach should be applied to reduce lung injury during the first few minutes of life. One of these interventions is the use of inspired oxygen in the delivery room (DR), which may contribute to acute lung injury, morbidity, and mortality.1,7
The survival rate of preterm infants has dramatically increased in the past 50 years but with no significant improvements in survival rate without morbidity in the last 10 years.8,9 Equipment used in the DR during newborn resuscitation has not kept pace with the equipment in the NICU environment. Initial monitoring is usually based on clinical examination alone and can be subjective.10 The strategies that are used in the NICU are not always applied in the DR. Care of the smallest preterm infants in the DR has received very little attention until recently. This is now changing as demonstrated by the 2006 addition of a dedicated chapter to the preterm infant in the recent edition of the Neonatal Resuscitation Textbook.11 To make a significant improvement in the transition from the fetal stage to birth and minimize the risks of serious morbidity related to the first few minutes of life, we should duplicate the technology and principles that are the new standard in the NICU.
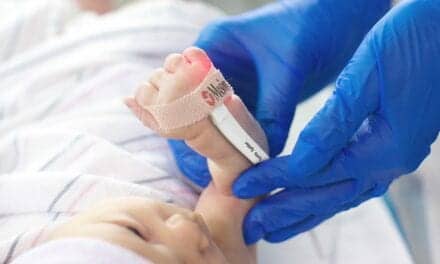
One technology that has dramatically improved care in the DR is use of pulse oximetry monitoring. Basing care of the newborn on the infant’s color is both inaccurate and imprecise.12 Preductal oximetry readings with accurate heart rate can be obtained from a newborn quickly. A stable heart rate would be an indication of a successful transition to the extrauterine environment. This could also provide continuous monitoring of heart rate during resuscitation efforts without stopping to obtain a heart rate via auscultation.
Utilizing oximetry to monitor oxygen saturation has limitations in clinical application. Currently, there is no standard for transitional saturations, although in utero acceptable transcutaneous oxygen saturation is approximately 60% and can drop as low as 30% during labor and delivery.13 Dawson et al and Rabi et al indicate that peripheral oxygen saturation (SpO2) of 60% immediately after birth and rising to >90% over 10 minutes is acceptable. At 5 minutes, some noncompromised infants may present with a SpO2 as low as 70%.14,15
At CHOC Children’s, blenders in the delivery room have become the standard of care, with the recommendation to start oxygen support with a FiO2 <40%. We titrate to maintain oxygen saturations between 85% and 92%, with incremental changes in FiO2. We tolerate lower SpO2 until after the 10-minute Apgar score is obtained.
Oxygen Monitoring
It has long been recognized that excess oxygen could cause permanent cicatricial retinopathy of prematurity as demonstrated in a collaborative controlled trial by Kinsey in 1956.1 Many studies have attempted to define what should be a safe level of arterial oxygenation. Both indwelling arterial lines and sensors have been used to monitor arterial oxygen tension, but no controlled trial had shown that monitoring alone has reduced the risk of permanent retinal damage until Flynn et al conducted a cohort study between 1982 and 1984 (published in 1992) in which they demonstrated the severity of ROP correlated with the number of hours with TcPO2 ≥80 mm Hg during weeks 1 to 4 of life.16 With this information, the use of pulse oximetry has become a standard of care in all neonatal centers. Initially, there were no guidelines for target ranges or alarm settings.
In 2001, Tin et al conducted a study with four centers that used different SpO2 alarm limits. In this study, they demonstrated that the lower the alarm ranges, the lower the incidence of ROP. The rate of ROP occurrences was 6% when the alarm settings were 70% to 90%, compared to 27% ROP occurrence with alarm settings of 88% to 98%.17 After the alarm ranges have been identified by any institution, the challenge becomes implementation. At CHOC Children’s, our current alarm settings are 85% to 92% for infants ≤1,500 g. Monthly audits reflect a variable compliance rate. Some months we have a 95%+ level of compliance, while in other months we have less than 85%. This has been an ongoing quality improvement project. A recent survey conducted by Ngheim et al demonstrated that even when SpO2 alarm-limit policies were in place, only one third of NICU nurses were aware of and able to identify their hospital’s policy.18 In addition, there is a low compliance in setting the alarm limits. Clucas et al demonstrated that when the low alarm target was 85%, compliance with policy was 95%, but when the upper limit target was 94%, compliance was 24%, and when the upper limit was 95%, compliance was 17%. An upper alarm limit of 96% had a 22% compliance rate. This demonstrates that patient care compliance has a direct impact on achieving the target oxygen levels.19 Another factor to consider is that even when the alarms are set in compliance with the institution’s policy, the patient can spend a high percent of time outside the target ranges. The alarm settings are broader than the target ranges. For instance, at CHOC Children’s, we target SpO2 85% to 92% but set our limits 80% to 97%. This is supported by a study from Hagadorn et al in which patients spent 16% of monitored time below the target range, 48% within, and 36% above the target range.20
Closed-loop Oxygen Management
Grodin et al analyzed a digital simulation of the respiratory control system. It demonstrated the role of central chemoreceptors and the effect of O2 and CO2 interactions on peripheral chemoreceptors.21 Sano described a closed-loop relation between inspired oxygen and arterial oxygen tension in a steady-state respiratory condition.22
A closed-loop-control delivery system is based on an adaptive or servo-controlled delivery system as opposed to an open-loop system used in current clinical practice in the NICU setting. The concept of a closed-loop system was investigated by Bhutani et al. In a clinical trial, he compared manual control (adjusted by a physician) and the software-based adaptive system using SpO2 as a feedback signal. FiO2 was titrated to a hood on nonventilated infants.23 His findings demonstrated that the desired SpO2 was achieved 81% of the time with the adaptive control as opposed to 69% of the time with a dedicated (physician) clinician at bedside. He also identified that the SpO2 target was achieved 54% of the time with the standard practice in the NICU. A similar trial was conducted by Urschitz et al on noninvasive nasal continuous positive airway pressure (NCPAP),24 and Claure et al conducted a similar clinical trial on mechanically ventilated very low birth weight infants.25 In both clinical trials, the adaptive controlled groups maintained a higher percentage of time at the target levels.
The most recent study on adaptive oxygen delivery was by Claure et al in the NICU at the University of Miami/Jackson Memorial Medical Center. The trial was conducted using the Avea ventilator (Carefusion) with a built-in FiO2 adjustment function utilizing SpO2 feedback (Radical, Masimo Corp).
The findings in this trial were that the time with SpO2 within the intended range (88% to 95%) increased during the adaptive period compared to the routine care period by 10%. Additional data points showed a reduction in the time higher than the intended range (21%) including a significant reduction in time with SpO2 ≥98% (13%). Also reduced was the amount of time with SpO2 ≤70%. An occurrence of hypoxemia secondary to a decrease in tidal volume was not identified as quickly due to the automated response, however.26
This trial demonstrates that a closed-loop system can improve our ability to maintain targeted saturation. This can lead to decreased morbidity. It also showed that the automated system must be monitored by trained individuals who will be able to recognize base changes in the FiO2 requirements and follow through to determine if this is a physiologic or mechanical event.
Methods of FiO2 administration to the VLBW infant have not significantly changed over the years. The change has occurred in the identified goal of oxygen therapy and the availability of monitoring devices. Adaptive FiO2 use is being introduced at the bedside. There will be much to learn as we move forward with this technology, but we must not forget to utilize our assessment skills and critical thinking; to focus on consistency to improve the care of the newborn. The care we provide will have a long-term impact on patients, their families, and society.
Roy Ramirez, RRT-NPS, RPFT, is clinical educator, respiratory therapy services, and ECLS coordinator; Sam Frens, RRT-NPS, is shift lead; and Eric Schultz, DO, MPh, is with the Neonatology Division, CHOC Children’s, Orange, Calif.
References
- Kinsey VE. Retrolental fibroplasia: cooperative study of retrolental fibroplasia and use of oxygen. Arch Ophthalmol. 1956;56:481-543.
- Gladstone IM Jr, Levine RL. Oxidation of proteins in neonatal lungs. Pediatrics. 1994;93:764-68.
- Munkeby BH, Barke WB, Bjomland K, et al. Resuscitation of hypoxic piglets with 100% O2 increases pulmonary metalloproteinases and IL-8. Pediatr Res. 2005;58:542-8.
- Tsao P, Wei S, Su Y, et al. Placenta growth factor elevation in the cord blood of premature neonates predicts poor pulmonary outcome. Pediatrics. 2004;113:1348-51.
- Attar MA, Domm SM. Mechanisms of ventilator-induced lung injury in premature infants. Semin Neonatal. 2002;7:353-60.
- Jobe AH, Ikegami M. Mechanisms initiating lung injury in the preterm. Early Hum Dev. 1998;53:81-94.
- Davis PG, Tan A, O’Donnell CP, Schulze A. Resuscitation of newborn infants with 100% oxygen or air: a systematic review and meta-analysis. Lancet. 2004;364:1329-33.
- Fanaroff AA, Stoll BJ, Wright LL, et al. Trends in neonatal morbidity and mortality for very low birthweight infants. Am J Obstet Gynecol. 2007;196:147.el-8.
- Halvorsen T, Skadberg BT, Eide GE, Roksund OD, Markestad T. Better C. Care of immature infants: has it influenced long-term pulmonary outcome? Acta Paediatr. 2006;95:547-54.
- O’Donnell CP, Gibson AT, Dacis PG. Pinching, electrocution, ravens’ beaks and positive pressure ventilation: a brief history of neonatal resuscitation. Arch Dis Child Fetal Neonatol Ed. 2006;9:F369-73.
- Kattwinkel J, ed. Neonatal Resuscitation Textbook. 5th ed. Elk Grove Village, Ill: American Academy of Pediatrics and American Heart Association; 2006.
- O’Donnell CP, Kamlin CO, Davis PG, Carlin JB, Morley CJ. Clinical assessment of infant colour at delivery. Arch Dis Child Fetal Neonatal Ed. 2007;92:F465-7.
- East CE, Colditz PB, Begg LM, Brennecke SP. Update on intrapartum fetal pulse oximetry. Aust NZ J Obstet Gynaecol. 2002;42:119-24.
- Dawson JA, Davis PG, O’Donnnell CP, Kamlin CO, Morley CJ. Pulse oximetry for monitoring infants in the delivery room: a review. Arch Dis Child Fetal Neonatal Ed. 2007;92:F4-7.
- Rabi Y, Yee W, Chen SY, Singhal N. Oxygen saturation trends immediately after birth. J Pediatr. 2006;148:590-4.
- Flynn JT, Bancalari E, Snyder ES, et al. A cohort study of transcutaneous oxygen tension and the incidence and severity of retinopathy of prematurity. N Engl J Med. 1992;326:1050-4.
- Tin W, Milligan DWA, Pennefather P, Hey E. Pulse oximetry, severe retinopathy, and outcome at one year in babies of less than 28 weeks gestation. Arch Dis Child Fetal Neonatal Ed. 2001;84:F106-10.
- Nghiem TH, Hagadorn JI, Terrin N, Sykes S, MacKinnon B, Cole CH. Nurse opinions and pulse oximeter saturation target limits for preterm infants. Pediatrics. 2008;121:e1039-46.
- Clucas L, Doyle LW, Dawson J, Donath S, Davis PG. Compliance with alarm limits for pulse oximetry in very preterm infants. Pediatrics. 2007;119:1056-60.
- Hagadorn JI, Furey AM, Nghiem TH, et al. Achieved versus intended pulse oximeter saturation in infants born less than 28 weeks’ gestation: the AVIOx study. Pediatrics. 2006;118:1574-82.
- Grodins FS, Buell RJ, Bart AJ. Mathematical analysis and digital stimulation of the respiratory control system. J Appl Physiol. 1967;22:260-76.
- Sano AMK. Adaptive control of arterial oxygen pressure of newborn infants under incubator oxygen treatment. Proc IEEE. 1985;132:205-211.
- Bhutani VK, Taube JC, Antunes MJ, Delivoria-Papadopoulos M. Adaptive control of the inspired oxygen delivery to the neonate. Pediatr Pulmonol. 1992;14:110-7.
- Urschitz MS, Horn W, Seyfang A, et al. Automatic control of the inspired oxygen fraction in preterm intants: a randomized crossover trial. Am J Respir Crit Care Med. 2004;170:1095-100.
- Claure N, Gerhardt T, Everett R, Musante G, Herrera C, Bancalari E. Closed-loop controlled inspired oxygen concentration for mechanically ventilated very low birth weight infants with frequent episodes of hypoxemia. Pediatrics. 2001;107:1120-4.
- Claure N, D’Ugard C, Bancalari E. Automated adjustment on inspired oxygen in preterm infants with frequent fluctuation in oxygenation: a pilot clinical trial. J Pediatr. 2009;155:640-5.