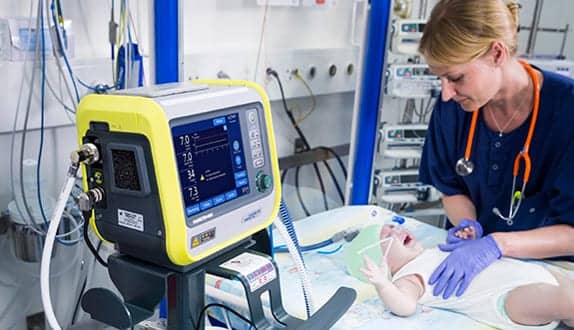
The HAMILTON?MR1 ventilator is now equipped with a CE-approved Neonatal Option, which means the device can now provide optimal ventilation therapy during MRI procedures for all patient populations, according to Hamilton-Medical.
The MR1 is FDA-approved and available in the US, however, the the Neonatal option is currently available only in the EU and EFTA member states and other countries that recognize CE marking, the company noted.
The HAMILTON-MR1 allows clinicians to take patients from the ICU to the MRI suite and back without having to change anything about the ventilation, even when they are on an advanced mode, the company says. This diminishes the risks for lung de?recruitment and a patient setback.
The new Neonatal option now also optimizes the HAMILTON?MR1 for MR?compatible neonatal ventilation. With tidal volumes as low as 2 ml, safe, and lung?protective ventilation of even the most fragile patients is guaranteed. The intelligent leak compensation function IntelliTrig automatically adjusts the inspiratory and expiratory trigger sensitivity to potential leaks. This enables optimal synchronization with the neonate’s breathing pattern, Hamilton says.
The company also says the HAMILTON?MR1 is the first ventilator able to be used at a magnetic field strength of 50 mT, equivalent to 1 m distance for a 3T static magnetic field scanner, without creating any MR image artifacts.
Positioning a medical device too close to the MRI scanner can have fatal consequences. For maximum safety, the HAMILTON?MR1 continuously monitors the magnetic field with TeslaSpy,an integrated gaussmeter, and gives you an audible and visual signal if you are getting too close.