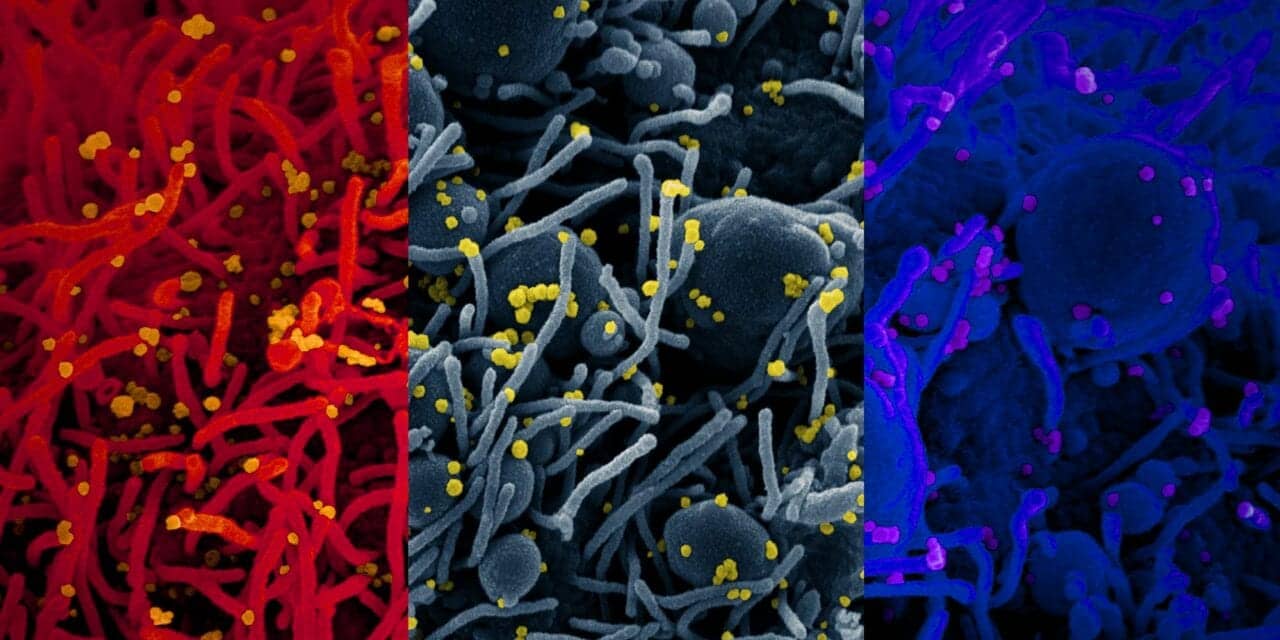
According to the National Institute of Allergy & Infectious Diseases (NIAID), three variants of the SARS-CoV-2 virus have been identified and are in circulation throughout the world:
- The variant (B.1.1.7 lineage) was first detected in the United Kingdom (UK) in September 2020;
- The variant (B.1.351 lineage) was first detected in October 2020 in the Republic of South Africa (RSA); and
- The variant (P.1 lineage) was first detected in Brazil in December 2020.
The NIAID says 47 countries (including the US) have recorded viral genetic sequences from cases involving the B.1.1.7 lineage (UK). In the United States, 293 cases involving the B.1.1.7 lineage have been reported in 25 states.
The B.1.351 lineage (RSA) has been recorded in 20 countries, but not yet in the US, according to NIAID.
Meanwhile, the P1 lineage (Brazil) has been found in five countries, with one reported case of P1 lineage in the United States, NIAID says.
“Viral variants are not unexpected, as all viruses evolve and mutate over time,” the NIAID said in a bulletin. “SARS-CoV-2 variants are a concern if they are more transmissible, cause more severe disease, or can evade immune responses to a greater extent than other SARS-CoV-2 strains, such that vaccines or antibody products are less effective.”
According to the NIAID, some scientific data suggest that the new variants may be more transmissible, prompting concerns that they could become more common in the population. Additionally, emerging data from the UK suggest that the B.1.1.7 lineage may cause more severe disease, but further confirmatory studies are needed, NIAID says.
A new variant evades the human immune response through a process called antigenic variation, whereby the virus changes its surface spike protein in a way that prevents antibodies from binding to and neutralizing the virus. Antigenic variation to SARS-CoV-2 is measured using known monoclonal antibodies to the spike protein on the virus surface or using sera from people who have either recovered from COVID-19 or who have been vaccinated with one of the FDA authorized COVID-19 vaccines currently available. Over the past several weeks, data have emerged to indicate that both the UK variant (B.1.1.7 lineage) and the RSA variant (B.1.351 lineage) are able to evade binding by some monoclonal antibodies to the spike protein.
In addition, studies of sera from individuals vaccinated with the FDA authorized mRNA COVID-19 vaccines suggest that the vaccines continue to induce a high level of neutralization when tested against the UK variants. When tested against the RSA variant, the mRNA vaccines induced a somewhat lower level of neutralization. “Although this decrease is of concern, the FDA authorized COVID-19 vaccines are expected to continue to protect against symptomatic and severe COVID-19 disease,” the NIAID said in a statement. “This is because these vaccines induce a more powerful immune response to SARS-CoV-2 than what results when someone is naturally infected.”
The NIAID said the vaccines’ 95% effectiveness against symptomatic COVID-19 and near-100% effectiveness against severe COVID-19 disease mean that even if these variants cause a modest reduction in the antibody levels generated by vaccination, “these vaccines should continue to provide a significant level of protection against illness.”
“Neutralizing antibodies generated by the FDA authorized COVID-19 vaccines are just one way that the immune system protects against severe disease. These vaccines also induce T-cell responses, which likely contribute protection against symptomatic and severe COVID-19 disease in concert with neutralizing antibodies,” the NIAID said.