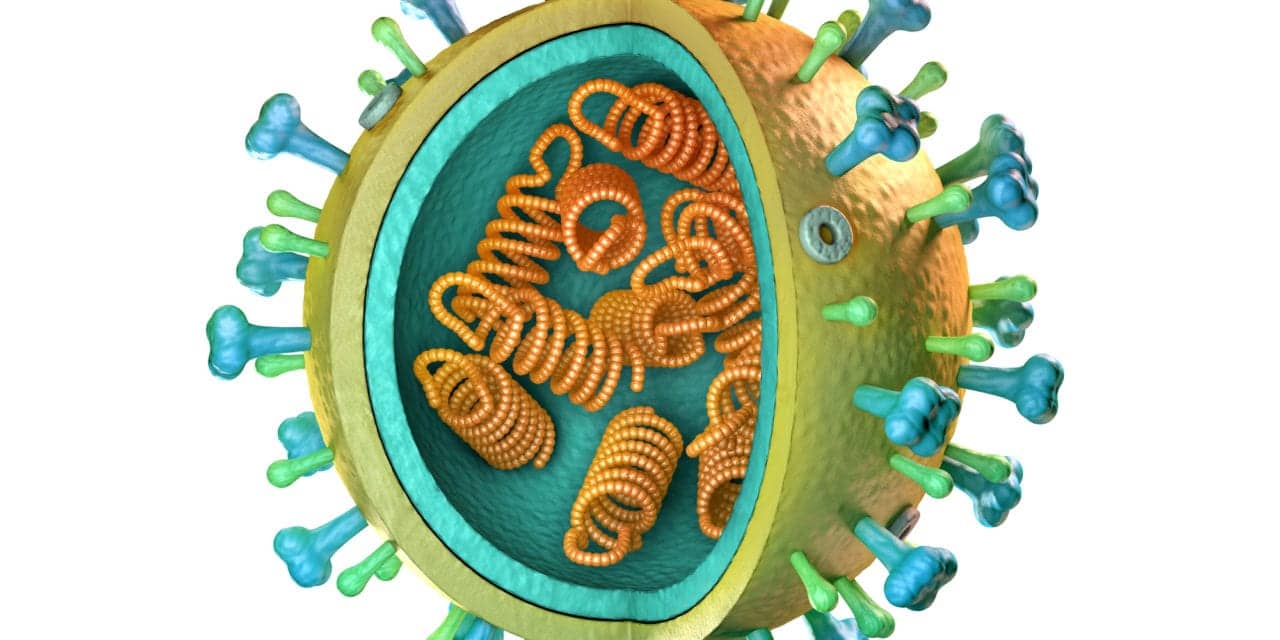
The clinical development program for the antiviral drug pimodivir is being discontinued since it failed to demonstrate a benefit over standard of care in certain severe influenza cases.
The efficacy and safety of pimodivir, an inhibitor of influenza virus polymerase basic protein 2, was being investigated in two phase 3 studies for the treatment of influenza A in hospitalized (ClinicalTrials.gov Identifier: NCT03376321) and non-hospitalized (ClinicalTrials.gov Identifier: NCT03381196) patients. Pre-planned interim analyses showed that among hospitalized patients with influenza A, pimodivir with SOC “was very unlikely to demonstrate added benefit”. As a result, the Company has halted both studies.
The decision to discontinue the pimodivir development program was reached in consultation with the Biomedical Advanced Research and Development Authority (BARDA), a co-funder for the program.
“While our goal was to develop an innovative new treatment option for patients at risk of respiratory infections, unfortunately these data show that pimodivir does not offer a benefit above the existing standard of care,” said James Merson, PhD, Global Therapeutic Area Head for Infectious Diseases at Janssen Research & Development.