A 41-year-old female admitted with diabetic ketoacidosis and possible sepsis benefited from prone positioning, which improved oxygenation in the later clinical course of acute respiratory distress syndrome.
Acute respiratory distress syndrome (ARDS) is a life-threatening clinical condition that increases mortality, lengthens hospital stays, and markedly consumes resources. Asbaugh et al1 first coined the term in 1967. The syndrome is now considered the most severe form of acute lung injury.2 The American-European Consensus Conference on ARDS3 recommends that acute lung injury be defined as a syndrome of inflammation and increased permeability that is associated with a constellation of clinical, radiological, and physiological abnormalities that cannot be explained by, but may coexist with, left atrial or pulmonary hypertension. ARDS consists of a group of clinical manifestations of evolving, severe, diffuse lung injury, especially of the lung parenchyma.
Multiple precipitating physiological derangements are associated with the development of ARDS. The reported prevalence and mortality rates for ARDS vary, with mortality reports ranging from 10% to 90%.4 Although there may be many reasons for divergent estimates, differences in diagnostic criteria, the variety of precipitating factors, and the lack of standardized tools for data collection add to the width of the range. It is estimated that about 150,000 cases of ARDS occur annually in the United States.5 Over the past 2 decades, many ventilatory and therapeutic modalities have been aimed at decreasing the ARDS mortality rate; these have included pressure-controlled ventilation, high positive end-expiratory pressure (PEEP), liquid ventilation, nitric oxide, extracorporeal membrane oxygenation, and prone positioning. Each of these modalities has been associated with limited success, and many are time-consuming, expensive, and available only in special institutions. Prone positioning is time-consuming, but is not expensive; it can be used in any institution.
P/F ratio.
Prone Positioning
Turning patients from the supine to the prone position has been proposed as a useful supportive therapy that can improve oxygenation in many patients with ARDS. Clinical studies6,7 of ARDS patients have reported that changing from the supine to the prone position improves oxygenation in 60% to 70% of patients and has minimal deleterious effects on hemodynamics. Almost 50% of patients maintained their improvements in oxygenation when they returned to the supine position.7 Theoretically, the most beneficial response to prone positioning would occur during the early, edematous phase of ARDS, when lung edema and atelectasis predominate. The acute phase of ARDS is characterized by damage to type I alveolar epithelial cells and impaired production and function of surfactant. Type I alveolar epithelial cells are responsible for gas exchange and help protect the alveoli from filling with fluid. Once these cells are damaged, the alveoli and interstitium quickly fill with blood and plasma, resulting in interstitial pulmonary edema.8 The chronic phase of ARDS is marked by thickening of the endothelium, epithelium, and interstitial space. Type I alveolar cells are destroyed and replaced by dysfunctional type II cells. Interstitial edema continues, resulting in fibrosis, and the alveoli continue to fill with protein-rich material that devastates the alveoli. Although these phases are well understood, the clinical feedback needed to define the critical time to institute prone positioning during the clinical course of ARDS has been lacking.9
Alveolar damage is widespread in ARDS, but CT indicates a disproportionately dependent distribution of radiographic density during the initial phase of ARDS. The reduction of these regional infiltrates in response to PEEP, as well as their rapid redistribution during changes of body position, suggests the potential for recruitment in those areas through increased transpulmonary pressure.10 When patients are supine, the abdominal contents exert pressure on the lower part of the diaphragm. Because pressure is greater on the dependent part of the diaphragm, the lungs are less inflated on inspiration. In contrast, the nondependent part of the lung is given a mechanical advantage; as a result, less inspiratory pressure is required for inflation. In the prone position, the pleural pressure gradient is reduced and the dorsal (nondependent) regions are exposed to a lower pleural pressure, resulting in the opening of previously atelectatic dorsal regions.11
Prone positioning evens the gradients of pleural and transpulmonary pressures, reshaping the lung’s contour and tending to recruit the previously dependent dorsal peridiaphragmatic regions. Because pulmonary blood flow remains distributed primarily to the dorsal quarters, better perfusion-ventilation matching is often accomplished.12 In addition, the weight of the heart in the prone position is borne primarily by the anterior chest wall, not by the dependent lung.13
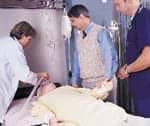 Post-resuscitation —clear lungs. |
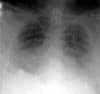 Post-resuscitation day 18, 24 hours prior to prone positioning. |
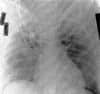 Twenty-four hours post-prone positioning. |
While many patients with ARDS experience improved oxygenation, not all patients respond (and, infrequently, deterioration is observed). Complications may occur either during or following patient repositioning. Careful attention to the endotracheal tube, chest tubes, and arterial and venous line can prevent their accidental removal during repositioning. Transient hemodynamic instability and oxygen desaturation may occur during repositioning. These can be minimized through adequate sedation and hyperoxygenation before turning the patient. Skin breakdown in weight-bearing areas (such as the face, chest, hips, and knees) can be minimized by using soft padding in the areas of contact, by using an inflatable bed, and by alternating between prone and supine positions every 8 to 12 hours.
Case Study
A 41-year-old female was admitted to Lehigh Valley Hospital, Allentown, Pa, on March 11, 2000, with diabetic ketoacidosis (DKA) and possible sepsis. She was treated with aggressive fluid management and other therapeutics using a DKA protocol. Her initial arterial blood gas analysis results indicated a pH of 6.99, a Paco2 of 10 mm Hg, and a Pao2 of 143 mm Hg. Her blood pressure was 80/40 mm Hg with an associated sinus tachycardia of 150 breaths per minute. After DKA protocol management, her diabetic ketoacidosis and hemodynamic status were stabilized and she began using low-flow supplemental oxygen. Several hours after admission, the patient suffered a cardiac arrest. She was resuscitated following Advanced Cardiac Life Support guidelines, but gastric aspiration occurred prior to intubation. After resuscitation, chest radiography revealed clear lungs and normal mediastinal structures.
The patient’s initial ventilator settings were 12 breaths of 600 mL per minute with a fraction of inspired oxygen (Fio2) of 50%, a peak inspiratory pressure of 38 cm H2O, a plateau pressure of 24 cm H2O, and a ratio of Pao2 to Fio2 of 255. Several hours later, the patient’s pulmonary compliance deteriorated, her Pao2:Fio2 ratio decreased to 100, and another chest radiograph demonstrated bilateral infiltrates consistent with ARDS. Despite aggressive ventilatory management, pharmacological paralysis, kinetic-bed therapy, and tracheostomy, the Pao2:Fio2 ratio remained near 100 and compliance was less than 20 mL/cm H2O.
Early in the course of pulmonary compromise, the use of prone ventilation was discussed, but it was not instituted due to the patient’s hemodynamic instability. Hemodynamic stability at this time was maintained by titrating doses of norepinephrine bitartrate and dopamine. Over the next several days, the patient’s oxygenation status remained critical, with an average Pao2:Fio2 ratio of 120, an Fio2 ranging from 60% to 90%, and PEEP levels of more than 12 cm H2O. Pulmonary compliance remained at less than 20 mL/cm H2O. Hemodynamic status slowly improved with aggressive fluid management and continued drug therapy. On the 19th day after the diagnosis of ARDS, the decision was made to attempt prone positioning. Prior to beginning prone positioning, the Pao2:Fio2 ratio was 60 and compliance was 18 mL/cm H2O. After 12 hours of prone positioning, the Pao2:Fio2 ratio improved to 120 and the Fio2 was reduced to less than 60% for the first time. Mucokinesis was enhanced, with the removal of thick secretions. Compliance remained unchanged. A chest radiograph taken 24 hours after starting prone positioning demonstrated a decrease in interstitial edema. Prone positioning was maintained for a total of 48 hours without any hemodynamic compromise. Improvement in skin integrity was also noted. The patient was returned to the supine position without any deterioration in pulmonary status. Subsequently, the Fio2 was reduced and the ventilator strategy was changed to pressure support in order to initiate the weaning process. Thirty-two days after admission, the patient was transferred to a weaning facility.
Conclusion
Turning critically ill patients from the supine to the prone position is an efficient method for improving gas exchange. It has been theorized that the maximum benefit from prone positioning would occur early in the clinical course of ARDS. This case, however, demonstrates that prone positioning can improve oxygenation in the later clinical course of ARDS. Even though the literature suggests that hemodynamic deterioration is limited during prone positioning, we believed that the patient’s oxygenation status and cardiac function were too volatile to attempt prone positioning earlier in the course of her illness.
It may be postulated, based on our clinical experience, that the stages of ARDS are not static; they may be dynamic. Since our patient responded to prone positioning with a noted improvement in the Pao2:Fio2 ratio and a reduction in pulmonary infiltrates, it appears that some degree of edema was present in the alveoli. Based on this case experience, the therapeutic use of prone positioning should be limited not by the clinical duration of ARDS but, rather, by clinical response.
Kenneth Miller, MEd, RRT, is a clinical educator, Lehigh Valley Hospital, Allentown, Pa. Angela Lutz, RRT, and Michelle Durnisch, RRT, are staff therapists at the hospital.
References
1. Asbaugh DG, Bigelow DB, Petty TL. Acute respiratory distress syndrome in adults. Lancet. 1967;2:319-323.
2. Andreadis N, Petty TL. Adult respiratory distress syndrome: problems and progress. Am Rev Respir Dis. 1985;132:1344-1346.
3. Bernard GR, Arrtigas A, Bringham KL, et al. The American-European Consensus Conference on ARDS. Am J Respir Crit Care Med. 1994;149:818-824.
4. Luce JM. Acute lung injury and the acute respiratory distress syndrome. Crit Care Med. 1998;26:369-376.
5. Millberg J, Davis D, Steinberg K, Hudson L. Improved survival of patients with ARDS. 1983-1993. JAMA. 1995;273:306-309.
6. Baiagoori F, Blanch L. Prone position in acute respiratory failure. Critical Care Alert. 1997;8:38-40.
7. Chatte G, Sab J, Dubois J, Sirodot M. Prone position in mechanically ventilated patients with severe acute respiratory failure. Am J Respir Crit Care Med. 1997;155:473-478.
8. Balas M. Prone positioning of patients with ARDS. Critical Care Nurse. 2000;20:24.
9. Langer M, Mascheroni D, Marcolin R, Gattinoni L. The prone position in ARDS patients. Chest. 1998;94:103-107.
10. Pappert D, Rossaint R, Slama K. Influence of positioning on ventilation-perfusion relationships in ARDS. Chest. 1994;106:1511-1516.
11. Broccard A, Shapiro S, Ravenscraft S, Marini J. Influence of prone position on the extent and distribution of lung injury in oleic acid model of ARDS. Crit Care Med. 1997;25:16-18.
12. Gattinoni L, Pelosi P, Vitale G, Presenti A, Mascheroni D. Body position changes redistribute lung computed-tomographic density in acute respiratory failure. Anesthesiology. 1991;74:15-23.
13. Albert RK, Leasa D, Sanderson M. Prone position reverses gravitational distribution of perfusion in dog lungs with oleic acid-induced injury. J Appl Physiol. 1990;68:1386-1392.