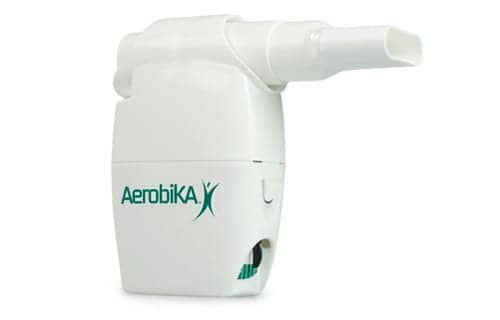
A new study presented at Chest 2016 showed Monaghan Medical Corp’s Aerobika device is effective in reducing drug use for treatment of COPD. COPD is a major source of morbidity and mortality in the United States. The Center for Disease Control estimates that COPD medical visits, hospital admissions and lost time costs 36 billion dollars annually. The recommended course of treatment for acute COPD exacerbations includes antibiotics and oral corticosteroids (OCS).
The study results showed that those study patients who used the Aerobika device experienced a significant reduction (57% and 89% reduction, respectively) in the use of antibiotics and OCS in the hospital setting compared to those study patients who did not use the device in addition to the regular COPD medication treatment. These findings are part of a larger 6-month retrospective study that demonstrated a 28% reduction in exacerbations in as little as 30 days of treatment, when used as an add-on to usual COPD medications.
This study showed for patients in the Aerobika device cohort, antibiotics were used 57% less and oral corticosteroids were used 89% less than for the cohort without the Aerobika device within 6 months’ post-exacerbation. The decreased need for short-term drug therapies including antibiotics and OCS, may have reflected better disease control with those patients who used the non-drug device. Additionally, patients in the Aerobika device cohort exhibited significantly lower costs throughout the study period with an average reduction of $6,347 USD and $9,936 USD per patient at 30 days and 6 months respectively for all in-patient and out-patient hospital costs.
“One of our major goals in developing the Aerobika device was to safely improve patient outcomes”, said Dominic Coppolo, MBA, RRT, FAARC, vice president Clinical Strategy and Development at Monaghan Medical. “These real-world findings are encouraging as we continue to conduct additional studies to further demonstrate the impact of our device in this high risk patient population.”










What about the real reason Patients are not doing well with their prescribed meds…they do not know how to use their INHALERS properly, and most have at least 2 or 3 INHALED MEDS AT HOME. Patient education is very inexpensive compared to readmissions and use of costly new
delivery devices.